Table 1. Summary of major imaging techniques
| Imaging technique | Features | Notes | General references |
|---|---|---|---|
| Wide-field | Limited image quality due to contribution of out-of-focus blur from above and below the focal plane | Most useful with thin tissues or culture cells or with low-NA, low-magnification dry objectives | Murphy (2001); Davis (2000); Wallace et al. (2001) |
| Deconvolution wide-field (3D imaging) | Post-image acquisition processing delivers increased signal-to-noise, resolution, and contrast. Requires multiple z sections | The most commonly applied imaging technique for high-resolution analysis of structure and dynamic processes (Davis 2000) | Wallace et al. (2001) |
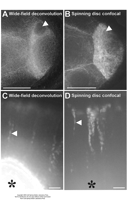
| Deconvolution wide-field (2D imaging) | Limited deblurring or deconvolution approaches applied to 2D xy image data to sharpen image detail | Useful increase in signal to noise and contrast when speed requirements/tissue sensitivity preclude z stack collection (see Comparison of confocal and wide-field images of stage-4 syncytial blastoderm embryos expressing GFP in the nuclei and Comparison of wide-field deconvolution and spinning-disc confocal images) | Parton and Davis (2004) |
| Point scanning confocal (or LSCM) | An optical technique that eliminates the contribution of out-of-focus light to the image to produce sharp “optical sections.” Alternative to deconvolution for elimination of out-of-focus blur | Useful in brightly labeled thick, hazy, or scattering material. Very good for high-resolution structural studies but suffers from speed limitations and its potential to affect biological processes (see Comparison of confocal and wide-field images of stage-4 syncytial blastoderm embryos expressing GFP in the nuclei and Comparison of confocal and multiphoton images of the same stage-4 syncytial blastoderm embryos expressing GFP in the nuclei) | Pawley (1995); Swedlow et al. (2002) |
| Spinning-disc or multifocal confocal | Similar to above but by using simultaneous multiple excitation beams has advantages in speed of image capture and fewer problems of dye photobleaching and phototoxicity | Increasing in popularity as an alternative to wide-field deconvolution for rapid dynamic processes (see Comparison of wide-field deconvolution and spinning-disc confocal images) | Diaspro (2001); Pawley (1995) |
| Multiphoton | Optical sectioning by the principal of multiple low-energy photon (700-1100 nm) absorption that occurs at extremely high illumination intensity, limiting dye excitation to an ~1-μm-thick focal plane | Promises the ability to image deeper in thick, hazy, or scattering material with improved cell viability. Limited application to Drosophila so far (see Comparison of confocal and multiphoton images of the same stage-4 syncytial blastoderm embryos expressing GFP in the nuclei; also see Movie 1 in Selection of Appropriate Imaging Equipment and Methodology for Live Cell Imaging in Drosophila). | Diaspro (2001); Amos (2000) |
Figure 1.
Comparison of confocal and wide-field images of stage-4 syncytial blastoderm embryos expressing GFP in the nuclei

Figure 3.
Comparison of confocal and wide-field images of stage-4 syncytial blastoderm embryos expressing GFP in the nuclei
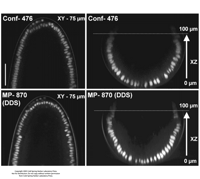
Figre 4.
Comparison of confocal and multiphoton images of the same stage-4 syncytial blastoderm embryos expressing GFP in the nuclei