Planting, Pollinating, Harvesting, and Monitoring Growth in Maize (Zea mays) for Research
- Zachary B. Traylor1,
- Sarah L. Fitzsimmons1,
- Melissa A. Draves2,
- Miriam Nancy Salazar-Vidal2,
- William F. Tracy3 and
- Sherry Flint-Garcia4,5
- 1Division of Biological Sciences, University of Missouri, Columbia, Missouri 65211, USA
- 2Division of Plant Science and Technology, University of Missouri, Columbia, Missouri 65211, USA
- 3Department of Agronomy, University of Wisconsin-Madison, Madison, Wisconsin 53706, USA
- 4Plant Genetics Research Unit, U.S. Department of Agriculture-Agricultural Research Service, Columbia, Missouri 65211 USA
- ↵5Correspondence: sherry.flint-garcia{at}usda.gov
Abstract
Zea mays, also known as maize or corn, is a staple crop as well as a classical model organism for plant genetic studies and research. To conduct maize research, plants must be properly cultivated in field or greenhouse conditions to ensure reproductive success and safeguard genetic identity through controlled pollinations. Genetic studies require knowing which alleles or genetic combinations (genotype) are present in an individual so the geneticist can create new combinations or select the desired genotypes. In order to determine and maintain the genetic identity of a corn plant and make precise selections of male and female plants, reproductive structures must be covered and isolated prior to silking and anthesis, or pollen shed. Doing so allows experimenters to make controlled pollinations to produce the desired genotype. Successful pollination of corn requires proper field design and preparation, careful planting to maintain distinct genetic families, and careful monitoring of growth and husbandry followed by proper harvest and seed storage. These activities have been optimized over the past 100 years. In this review, we summarize each step needed to produce a generation of corn from planting to harvest.
INTRODUCTION
Zea mays ssp. mays, also known as maize or corn, is a primary driver of the current agricultural economy in the United States and has been a model for plant genetics studies for >100 years (Agarwal et al. 2018). Geneticist George Beadle (1939) first hypothesized that teosinte (Zea mays spp. parviglumis) was the ancestor of maize. Over thousands of years, humans modified a range of domestication and productivity traits in corn plants, starting with teosinte and ending in modern maize. Initially, humans selected for traits associated with easier harvest and against distinctive teosinte traits, which resulted in a maize-like plant with fewer tillers (lateral branches originating at ground level) and fewer ears per plant, each of which bore “naked grains” that lack the hard outer glume (which are leaf-like structures in maize) present in teosinte (Doebley 2004). After the initial domestication, native peoples carried early maize across the Americas and this early maize adapted to new environments (Yang et al. 2023). These locally adapted populations are known as landraces and were grown by farmers until the early 1900s, when initially public sector (universities and the United States Department of Agriculture [USDA]) and subsequently private sector (seed industry) breeders selected agronomically successful landraces, ultimately resulting in the modern maize hybrids that we know today. Understanding this history of maize is important to appreciate the diversity in varieties, plant structures, and development discussed herein.
Conducting maize research requires proper cultivation and controlled pollinations. Over the years, there have been many protocols on growing, pollinating, and harvesting corn published in various books and journals by eminent leaders in maize genetics (Kiesselbach 1949; Wallace and Bressman 1949; Sheridan 1982; Freeling and Walbot 1994). As part of this collection, we provide updated, comprehensive methods, visuals, and troubleshooting guides for these protocols to support maize genetics and breeding research. Select methods are applicable across multiple environment types, including the field, greenhouse, and growth chamber, as well as for field arrangements including nursery, genetic experiments, breeding, or phenotypic trials. This review first provides background on maize structure, development, and terminology, followed by a description of how and why controlled pollinations are carried out. Growth environments for maize experiments are discussed. The review then turns to introducing our associated protocols, which are focused on: planting (Draves et al. 2025), pollinating (Fitzsimmons et al. 2025), harvesting (Salazar-Vidal et al. 2025a), and monitoring growth and developmental stages (Salazar-Vidal et al. 2025b). Each of these protocols is critical to the success of maize experimentation and ultimately to increasing basic scientific knowledge and continued improvement of the maize crop.
MAIZE STRUCTURE AND DEVELOPMENT
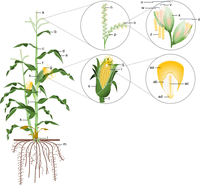
Maize is an annual, monoecious plant with tassels (male reproductive organs) and ears (female reproductive organs) that are separated on the plant, making maize a natural cross-pollinator (Fig. 1; Neuffer 1994). The physical separation allows for easy and reliable controlled pollinations, thus making it a suitable model for classical genetic studies. Humans have adapted maize varieties to nearly every environment in which humans have introduced agriculture. As such, plant size and rate of development vary widely, with some landraces that complete a generation (from the planted seed to mature progeny seed) in 2 months, while other landraces require 10 months. However, the basic morphology and stages of development are conserved. The maize stalk (or stem) consists of nodes separated by internodes (Fig. 1). At each node a single leaf is initiated, and a reservoir of meristematic cells may or may not develop into additional structures, depending on the type of maize and the growing environment. Below ground, these meristematic cells form the primary root system. Meristems below ground may also develop into lateral branches called tillers. Above ground, these meristematic cells may develop brace roots or prop roots at several nodes near the ground. After the brace root nodes, these reservoirs of meristematic cells along the main stalk are usually suppressed until the middle of the plant, where they develop into lateral branches tipped by ears. Above the branches tipped with ear, the meristems are suppressed again until reaching the uppermost node on the stalk that develops into the tassel.
Anatomy of a maize plant, its reproductive structures, and the kernel. Maize structures include (a) tassel, (b) uppermost leaf, (c) stalk or stem, (d) leaf blade, (e) midrib, (f) leaf sheath, (g) silks, (h) primary or top ear, (i) secondary ear, (j) node, (k) tiller, (l) brace roots, (m) root system, (n) main spike, (o) spikelet pair, (p) tassel branch, (q) cob, (r) kernel, (s) husk leaf, (t) shank or lateral branch, (u) open pedicellate spikelet, (v) open upper floret, (w) closed lower floret, (x) glume, (y) closed sessile spikelet, (z) anther, (aa) endosperm, (ab) scutellum, (ac) plumule or embryo, and (ad) pedicel.
Maize starts to emerge from the ground at germination (the VE, or emergence, stage) as a cotyledon with a single leaf. After emergence, the plant grows, adding additional leaves and, eventually, clearly defined reproductive structures. Stages of plant growth are assigned V for vegetative development and R for stages of reproduction (Richie et al. 1986; Salazar-Vidal et al. 2025b, in this collection). The V number is determined by counting the number of visible leaf collars, which are the structures where the leaf blade attaches to the leaf sheath, and typically ranges from V1 to V20 (Richie et al. 1986). The R1 designation is given when silks emerge, and subsequent R designations are based on the developmental stage of the developing kernels. Around R1, pollen is shed from the tassel, and the silks receive pollen and guide the developing pollen tube to transfer sperm to the ovule for fertilization and kernel development. After pollination, the primary focus of the plant shifts to kernel development. When the kernels are fully mature, the plant dies, and the ears can be harvested to allow for the propagation of the next generation. In total, the entire life cycle of a maize plant from planting to harvest takes 4–5 months to complete, although there is wide variability in this time frame.
MAIZE TERMINOLOGY
In plant research, the terminology identifying a specific group of individuals (i.e., line, cultivar, variety, etc.) is variable and specific to the model organism and the literature of that species. In maize, the terms “line” and “population” are typically used. Lines refer to more uniform materials with similar or identical genetics, while a population is used to identify groups of genetically related individuals, or a group of unrelated individuals. Here we explain some of this terminology.
In modern maize production, farmers typically grow hundreds or thousands of acres of the same variety. The type of corn planted in these large fields is a hybrid, which is produced by crossing two inbred lines. In maize research, inbred lines (inbreds) are varieties that have been developed by successively self-pollinating (selfing) for eight or more generations, creating a very high level of homozygosity and uniformity. Developing inbred lines is a major goal for breeders. However, inbred lines are low-yielding; thus, a second major breeding goal is creating and identifying high-yielding hybrids by crossing inbred lines together to produce heterosis or hybrid vigor.
In genetics studies, inbred lines are typically used for genetic screens or when a researcher wants a consistent genetic background. Examples of inbred lines include B73, Mo17, and W22. There are tens of thousands of inbred lines in the public sector alone (Gerdes et al. 1993), each with distinct genetic, physical, molecular, and chemical characteristics in all parts of the plant.
In maize research, the term “population” is used in numerous ways. First, a population can be a group of related individuals. For example, if two inbred lines are crossed together to make an F1 hybrid and the hybrid is self-pollinated, the progeny generation is a population of F2 individuals that can be used to map mutants or quantitative trait loci (QTLs). The F2 individuals can be self-pollinated to create a population of F2:3 families (in other words, F3 families each derived from a single F2 individual), which can also be used to map genes that underlie traits. Inbreeding can continue for five or six generations (depending on the acceptable level of heterozygosity: 3.1% heterozygous after five generations of selfing or 1.6% heterozygous after six generations of selfing), which results in a population of recombinant inbred lines, a genetically stable population that is no longer segregating. Another example of how the term “population” is used is a breeding population, where two or more parents are crossed to bring together desirable traits. A breeding population can be mated several times to make new combinations and then self-pollinated to make new inbred lines and future hybrids. A third meaning of the term “population” is a collection of lines that may be unrelated, where characterizing genetic diversity is key. Association populations are composed of hundreds of unrelated inbred lines, which are again used to map QTLs. Examples include the 282 Association Panel (Flint-Garcia et al. 2005), the Wisconsin Diversity Panel (Hansey et al. 2011), and the Ames Panel (Romay et al. 2013). A fourth meaning of “population” includes mutant populations, where mutations are created in a specific inbred background by either transposon insertions (e.g., BonnMu) (see Marcon et al. 2024, in this collection) or chemical mutagenesis (e.g., EMS, ethyl methanesulfonate) (see Khangura et al. 2025, in this collection). Finally, landraces and teosintes are typically heterogeneous populations that do not tolerate inbreeding and must be maintained as a noninbred population of related individuals, often referred to as an “accession,” which is a sample of seeds collected from a specific place at a specific time.
The term “germplasm” refers to the entire collection of genetic material for a plant species; in the case of maize, germplasm is maintained as seed. In the United States, two of the most used germplasm collections maintained by the USDA National Plant Germplasm System include the Maize Stock Center (http://maizecoop.cropsci.uiuc.edu/), which houses seeds of mutant collections and mapping populations, and the North Central Region Plant Introduction Station (https://www.ars-grin.gov/), which houses seeds of wild relatives, inbred lines, and breeding populations. Each country has their own germplasm collections, as do private companies.
GROWTH ENVIRONMENTS FOR MAIZE
Once lines or populations have been developed in nurseries, growing maize as described in this review also applies to experiments where the purpose is data collection, selection, and inbred line development (Gaffney et al. 2015). There are a variety of growth environments that can be used for conducting experiments. Preferably, maize should be grown outside in the field, as it is a large plant that requires a lot of space and soil. For small experiments involving tissue collection, germination, or mutant scoring, maize can be grown in a greenhouse or growth chamber. Greenhouses or off-season winter nurseries can also be used for growing maize beyond the typical growing season to advance another generation before the following summer. Winter nurseries in tropical, day-neutral sites with 12 h day lengths, such as Mexico, Hawaii, and Puerto Rico, offer the benefit of a compressed/shortened growth season that can be used to accelerate maize breeding programs for laboratories found at higher latitudes. However, winter nurseries require specialized facilities and travel expenses, and thus can be substantially more expensive than local, summer nurseries. Winter nurseries in the southern hemisphere, such as in southern Chile, offer growth conditions similar to the temperate regions of the United States. Typically, the turnaround time between fall harvest after the 4–5 month summer field season in the United States and winter nursery planting is quite short, so prompt harvest and processing and early planning are imperative.
Various field arrangements can be used, depending on the amount of maize to be grown and the amount of controlled pollination required. For genetics studies, the most common field arrangement is a nursery, where families of related plants are grown in rows and can be self-pollinated, crossed within the family, or crossed with other germplasm. Genetics nurseries are smaller fields, typically up to an acre, and are particularly helpful for making specific crosses and scoring populations to select plants for advancement to the next generation. For this reason, genetics nurseries are the best arrangement for forward and reverse genetic studies to identify target phenotypes, and for early phases of population development. Advanced planning on spatial organization of rows and on planting quantities for future nursery and experimental needs is key to maximizing field space and efficiency in a nursery setting. A second common field arrangement is the experimental plot, where the main purpose is to evaluate the germplasm. Such an arrangement is usually focused on a testable hypothesis. Experimental plots typically have blocks of replicated material to test the question of interest and include control or check plots (plots containing a control genotype to standardize data between different sections of the field). Experimental plots can be used for testing specific environmental conditions (e.g., drought vs. well-watered), assessing populations in the field for diseases or insect resistance, or comparing phenotypes of mutated lines. Researchers often use experimental plot arrangements for QTL mapping and association mapping for agronomic traits such as germination, disease, and overall plant health in large populations (Badu-Apraku et al. 2012). The final common field arrangement is a yield trial field. These large field trials typically involve machine planting to ensure uniformity and are open-pollinated (not manually pollinated) because the seed is not saved for future studies. The primary focus of yield field trials is collecting agronomic phenotypes and allowing each plant to reach its full yield potential. Because performing manual pollinations would interfere with plant and ear development and therefore limit yield, it is not performed in yield studies.
METHODS FOR PLANTING, POLLINATING, HARVESTING, AND MONITORING GROWTH
Successful planting begins with planning. Planning a field before a study begins should be a part of experimental design when data are to be collected, and also in nurseries, to arrange families in close proximity for convenience of cross-pollination. Field design and staggering planting dates will also aid in lining up flowering dates of different populations for optimal crossing. Field design may also be useful for organizing blocks for breeding efforts using the same logic for crossing; e.g., blocking the self-pollinations together and the sibling pollinations together. In terms of planting, each improvement level of maize (landraces, older inbreds, and modern inbreds) has an optimal planting density, and should be planted as such to prevent barrenness (Tokatlidis and Koutroubas 2004). For small-scale research plots and nurseries, corn is commonly planted and harvested by hand. However, machine planting and machine harvesting may be necessary, depending on the goals and size of the research project. Care should be given to soil fertility; weed, insect, and disease management; and irrigation throughout the growing season.
For planting large field trials, such as yield trials or other large experiments where precision is required, a machine planter is typically used for planting. Planting with a machine allows for uniform spacing of plants within and between rows, as well as proper border rows around plots so that consistent growing conditions apply to all plots, and genetically complex traits such as yield are accurately measured. In the case of experimental plots, planting should also involve controls within a block to standardize the data across the field, especially where there is variation within the field in terms of soil quality or water availability. Additionally, the researcher must decide how to arrange the field using appropriate experimental designs (i.e., randomized block designs, split plot designs, etc.) (Badu-Apraku et al. 2012; Mohr et al. 2022). Once the seed is packaged for either a nursery or a trial, the packets are arranged in the proper order depending on whether the seed will be planted by hand by laboratory personnel or machine planted by trained personnel.
Plant growth is monitored throughout the growing season, regardless of whether it is a nursery, an experimental plot, or a yield trial. As highlighted in historical guides (Kiesselbach 1949; Richie et al. 1986), there are key developmental transitions that occur during maize growth, and understanding when they occur will aid in timing of experimental processes such as sample collection and data collection. Knowing when the plant transitions from juvenile to adult to reproductive stages of growth will inform the researcher when plants will flower and when to prepare for and conduct pollinations. After pollinations, careful monitoring of the transition to plant senescence and kernel physiological maturity is important for proper timing of harvest. There are additional specific time points in kernel development (Richie et al. 1986) that may be relevant for time-sensitive experiments or sample collection. For example, kernel protein accumulation begins ∼14 days after pollination (R2 stage) and starch accumulation is active ∼20 days after pollination (R3 stage). Special care should be taken if the experiment contains various germplasm with a wide range of maturities, as key developmental time points will be reached at different times in lines with different growth rates.
Once the plant reaches the reproductive stage, controlled pollination is critical to tracking the genetic identity and pedigrees of the progeny (Neuffer 1994). Because corn is a cross-pollinated species, pollen can travel via gravity, insects, and wind. Therefore, to prevent contamination of the ear by unwanted pollen, the ear must be covered with a shoot bag prior to silk emergence, and pollen must be collected from the desired tassel using proper pollination techniques (Neuffer 1994). If a plant is to be self-pollinated (selfed), pollen from the tassel of a plant is used to pollinate the ear of the same plant. If a plant is to be cross-pollinated (crossed), pollen from the desired tassel is used to pollinate the ear of a different genotype (Beal 1878). The precise steps of this process are described in detail in Fitzsimmons et al. (2025) in this collection, and using this method, a maize researcher can conduct a variety of experiments to fit their research goals. Whether the cross takes place within a family of related plants (a sibling cross) or between families, the same precautions are needed for shoot bagging and tassel bagging. Contamination is possible in all types of crosses as a result of an exposed silk prior to shoot bagging or during pollination, or from a tassel contaminated by pollen from nearby plants. Either case results in the incorrect genetic identity and will affect future generations, so good pollination techniques are essential. Alternatively, corn can be grown in isolation plots to allow open pollination to occur without the risk of pollen contamination from other plots. Isolation can be achieved by either (1) physical isolation, e.g., by planting at least 200 m away from other maize plantings, or (2) temporal isolation, where plantings are timed so the isolated population is not flowering at the same time as nearby corn (USDA-APHIS 2013).
Pollination techniques are fairly standard for most maize research programs. Geneticists use sibbing and selfing pollination methods to create families that are segregating for the genes of interest and to cross a mutation into different backgrounds to better visualize a mutant. Naturally occurring or artificially generated mutants (see Khangura et al. 2025, in this collection) often result in phenotypes that can be detected visibly, biochemically, or molecularly, and allow forward genetic studies, such as elucidating gene action by examining Mendelian segregation ratios and interactions with other known mutants and candidate genes (Candela and Hake 2008). The process of controlled pollination is also applicable for reverse genetic screens (Till et al. 2004; Weil and Monde 2007; Liu et al. 2020). In this instance, a target gene is mutated and then plants are grown for phenotype observation. Through controlled pollination, the progeny of the mutant plants remain stable for future studies.
Breeders use crosses within and between different inbred lines and populations to generate new parental populations and use self-pollination to develop inbred lines (Kiesselbach 1949; Harjes et al. 2008). Quantitative geneticists use the same pollination methodologies as breeders in their nurseries to create segregating populations and map quantitative traits (Lee et al. 2002; Flint-Garcia et al. 2005). Breeders or quantitative geneticists who want to maintain high levels of heterogeneity in landraces and other heterogeneous populations use sib pollination techniques to cross plants within the same family to avoid inbreeding depression. A variety of other controlled pollination strategies can be used to combine the genetics of different parents by crossing them together, randomly mating the progeny to generate recombination events, and self-pollinating the F2 progeny to generate inbred lines.
Controlled pollination is also required when evaluating grain traits where xenia is a concern. Xenia is the phenomenon where the genetics of the pollen influence the phenotype of the kernel; e.g., sweet corn pollinated by field corn pollen will result in nonsweet kernels. In contrast, most large field trials do not require manual pollination, and the plants are allowed to open-pollinate and produce uncontrolled progeny. In trials focused on plant growth, abiotic stress tolerance, or insect or disease resistance, the genetic identity of the progeny seed is not important.
Finally, timely harvest of mature seeds and proper storage are critical to maintaining genetic and breeding stocks across long periods of time, often >30 years under proper storage conditions. In a typical field season, planting will happen in late spring, allowing for an early fall harvest, ideally at physiological maturity, 40–50 days after pollination. Delayed harvest may result in insect, disease, or animal damage to the ears, as well as broken stalks as the plant senesces.
Harvesting time lines are largely dictated by the material to be harvested. With genetics trials, the use of a small number of common inbred lines means that harvest will likely occur within a short time frame as all plants flower, senesce, and are ready for harvest at the same time. The same is true for field trials containing large amounts of related germplasm grown for observation. Field trials focused on yield and yield component traits (e.g., ear length, kernel row number) should be designed early to determine which traits will be assessed and how and when the data will be collected, as there are a number of high-throughput methods available today (Badu-Apraku et al. 2012; Tandzi and Mutengwa 2020). In contrast, breeders and geneticists using more diverse germplasm will have greater variability in when their populations are ready for harvest. Special attention should be paid as to when each population begins to senesce and kernel maturity is reached (Richie et al. 1986) to best judge when to harvest the trial. It is also significant to note that harvesting, whether in nurseries or larger trials, is a dynamic process. Kernel type (dent, sweet, floury, etc.), local environmental conditions (temperature, rain, and wind), and whether the germplasm is adapted to your environment will alter the time frame when harvest should occur. For example, if disease is a common issue during early kernel maturity, or if late-season weather causes losses before the kernels are fully developed, it may be beneficial to harvest slightly earlier to save healthier seeds.
DATA ACQUISITION
Throughout the season, data can be collected starting at germination and continuing through harvest. Soon after planting, seedling emergence can be used to study seed viability of a particular seed lot, as well as for replanting if seeds do not germinate due to low temperature or low/excessive moisture conditions. As the plants develop, phenotypic data can be collected for any number of developmental and morphological traits. Tissue samples can be collected for biochemical or genetic analysis, including leaf samples for genotyping families or populations for both genetic studies and marker-based selection (Bernardo and Yu 2007; Gazal et al. 2015). Data can be collected for reproductive traits such as male sterility by recording whether plants shed pollen or not (Weider et al. 2009) and flowering dates. Between flowering and harvest, morphological data can be collected, including reproductive mutant phenotypes, mature plant height (Ku et al. 2015), disease and insect susceptibility, stalk and root lodging, and yield. Stalk lodging occurs when the stalk breaks at or below the ear, resulting in ear loss during mechanical harvest (Hu et al. 2013; Berry et al. 2021), while root lodging refers to the root system not being able to sufficiently hold up the plant and results in the whole plant leaning or falling (Fincher et al. 1985; Kamara et al. 2003; Berry et al. 2021). More recent advances in high-throughput genotyping have also yielded single-seed genotyping platforms that allow genotyping of seeds before planting, as opposed to collecting leaf samples during development (Xu and Crouch 2008), thus saving space in the nursery.
To study growth and development, researchers can manipulate both artificial and natural variables to test hypotheses related to their research of interest. Some of these conditions include designing drought studies using well-watered versus drought-simulated effects (Cooper et al. 2014; Gaffney et al. 2015; Danilevskaya et al. 2019), disease studies that require pathogen inoculation (Tegegne et al. 2008; Mesterházy et al. 2012; Sorensen et al. 2025, in this collection), insect studies that require infestation (Butron et al. 2018), and protocols outlined by the Animal and Plant Health Inspection Service (APHIS) for transgenic maize (https://www.aphis.usda.gov). However, for the purposes of this review, we focus on the core protocols of growing, pollinating, and harvesting maize under standard field growing conditions.
CLOSING REMARKS
Through this introduction, we have summarized the key aspects of a growing season for corn research. Corn is a classical genetic model due to its evolutionary history of being selected as a crop and its clear separation of reproductive organs. In tandem with describing the history of maize as a model, this introduction also has provided some terminology and resources that researchers can use to inform their own research questions. We also have outlined the importance of using controlled pollinations when appropriate and designing a growing season that best suits the needs of the researcher. Finally, we have provided an overview of each aspect of a growing season, from field design to harvest, that will be further elaborated on in our associated protocols (Draves et al. 2025; Fitzsimmons et al. 2025; Salazar-Vidal et al. 2025a,b).
COMPETING INTEREST STATEMENT
The authors declare no competing interests.
AUTHOR CONTRIBUTIONS
Conceptualization: Z.B.T., S.L.F., M.A.D., M.N.S.-V., and S.F.-G. Writing—original draft: Z.B.T. Writing—review and editing: S.L.F., M.A.D., M.N.S.-V., W.F.T. and S.F.-G.
ACKNOWLEDGMENTS
Work in our laboratory is supported in part by the U.S. Department of Agriculture (USDA), Agricultural Research Service. The findings and conclusions in this publication are those of the authors and should not be construed to represent any official USDA or U.S. Government determination or policy. Mention of trade names or commercial products in this publication is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the U.S. Department of Agriculture. The USDA is an equal opportunity provider and employer.
Footnotes
-
From the Maize collection, edited by Candice N. Hirsch and Marna D. Yandeau-Nelson. The entire Maize collection is available online at Cold Spring Harbor Protocols and can be accessed at https://cshprotocols.cshlp.org/.
REFERENCES
*Reference is also in this subject collection.